Like charges repel, whereas unlike charges attract. (i) Chemical reaction: Na Cl ) (e NaCl (1.1) )0 ( (ii) (iii) (iv) (v) Radioactive decay: A neutron will decay into a positively charged proton, a negatively charged electron and a neutral charged electron-anti-neutrino, n p e (1.There are only two types of charge, one called positive and the other called negative. Accurate knowledge about the proton charge radius is not only essential for understanding how quantum chromodynamics (QCD) works in the non-perturbative region, but also important for bound state quan- tum electrodynamics (QED) calculations of atomic energy levels.The effects of static electricity are explained by a physical quantity not previously introduced, called electric charge.Some of the most basic characteristics of static electricity include: Attendants in hospital operating rooms must wear booties with a conductive strip of aluminum foil on the bottoms to avoid creating sparks which may ignite flammable anesthesia gases combined with the oxygen being used. The neutrons carry no charge, and their number can vary. it is electrons that just move and decides positive and negative charge on body'. An atom is in balance when it has an equal number of protons and electrons. we do not use proton excess or deficiency terminologies because protons are way more heavier than electrons and do not move when we charge a body either negative or positive. Like charges repel, whereas unlike charges attract. 'deficiency of electrons results in positive charge on body, while excess of electrons means body have negative charge. There are only two types of charge, one called positive and the other called negative. Conversely, the charge of an electron is -1.6 x 10 -19 C. The charge on 1 proton is 1.6 x 10 -19 C. 
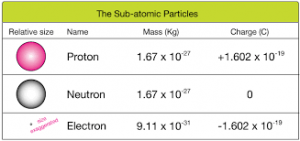
One coulomb is equal to the charge on 6.241 x 10 18 protons. One coulomb is equal to the amount of charge from a current of one ampere flowing for one second. When we pump gasoline, we are warned to discharge ourselves (after sliding across the seat) on a metal surface before grabbing the gas nozzle. Some of the most basic characteristics of static electricity include: The effects of static electricity are explained by a physical quantity not previously introduced, called electric charge. The coulomb, also written as its abbreviation C, is the SI unit for electric charge. We also have to be cautious of static electricity, especially in dry climates. You can also rub a balloon on your hair, and the static electricity created can then make the balloon cling to a wall. Similarly, lightning results from air movements under certain weather conditions. Static cling generated in a clothes dryer and the attraction of straw to recently polished amber also result from rubbing. Almost all of the mass of an atom is contained within a tiny (and therefore extremely dense) nucleus which carries a positive electric charge and almost all of the volume of an atom consists of empty space in which electrons reside (Figure 1.8. Rubbing creates the spark you get from walking across a wool carpet, for example. Many of the characteristics of static electricity can be explored by rubbing things together. (credit: Sebakoamber, Wikimedia Commons). If a body contains x number of electron and y number of protons, then the magnitude of charge on the body will be: (x - y) × 1.6 × 10-19 C (x y) × 1.6 × 10. At the same time, the silk, having lost electrons, becomes positively charged. Between them neutron does not have any electric charge but electron is negative and against that. When a piece of amber is rubbed with a piece of silk, the amber gains more electrons, giving it a net negative charge. Electron, proton and neutron are the most important Subatomic particles which made atoms. Charge Of Proton Proton, a stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10 27 kg, which is 1,836 times the mass of an electron. Both protons and neutrons have a mass of 1, while electrons have almost no mass. The negative charge of one electron balances the positive charge of one proton. \) electrons have a combined charge of −1.00 coulomb.\): Borneo amber was mined in Sabah, Malaysia, from shale-sandstone-mudstone veins. As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The elementary charge or proton charge e has the SI unit coulomb C, but more strictly coulomb per elementary unit Cx-1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
